Overvåkingsprogrammet for sykdommene bonamiose og marteiliose i flatøsters og marteiliose i blåskjell utføres av Havforskningsinstituttet på oppdrag fra Mattilsynet. Det ble i 2022 hentet blåskjell og flatøsters fra et område i Agder hvor det foregår både dyrking og kommersiell høsting av skjell, og ett blåskjellanlegg i Trøndelag. Prøvene ble samlet inn i april/ mai og oktober, når prevalensen av parasittene Bonamia spp. og Marteilia spp. er vist å være høyest i smittede bestander. Det er ikke observert unormal dødelighet verken vår eller høst. Bonamia ostreae / B. exitiosa ble ikke påvist. M. refringens er ikke påvist. En Marteilia sp. ble for første gang påvist i ville blåskjell, Mytilus edulis, på Bømlo i 2016, og det er gjennomført en kartlegging i 2022 som vil bli videre beskrevet i et pågående forskningsprosjekt. Marteilia er så langt påvist på åtte lokaliteter. Alle funn er sekvensert og typet som Marteilia pararefringens, som antas å være spesifikk for blåskjell. Det foreslås en revisjon av overvåkingsprogrammet og søknad om etablering av fristatus for Bonamia exitiosa i norske flatøsters. Videre foreslås søknad om oppheving av kontrollområdene for Bonamia ostreae og Marteilia refringens i hhv. Arendal og Bømlo.
The surveillance and control programme for bonamiosis and marteiliosis in European flat oysters, Ostrea edulis, and blue mussels, Mytilus sp. in Norway in 2022
Report series:
Rapport fra havforskningen 2023-25
ISSN: 1893-4536
Published: 01.06.2023
Project No.: 14538
On request by: Mattilsynet
Reference: Odd Arne Brimsøe
Research group(s):
Smittespredning og sykdom
Subject:
Flatøsters,
Blåskjell
Program:
Miljøeffekter av akvakultur,
Kystøkosystemer
Approved by:
Research Director(s):
Geir Lasse Taranger
Program leader(s):
Terje Svåsand
Norsk sammendrag
Summary
The surveillance programme is carried out by the Institute of Marine Research according to a contract with the Norwegian Food Safety Authority. In 2022, samples were collected from an area in Agder where flat oysters and mussels are harvested from wild beds and farmed, and from one mussel farm in Trøndelag county. Samples were collected in April/May and in October, to be able to detect Bonamia sp. and Marteilia sp. during the periods when the potential prevalence in affected populatioons is highest. No abnormal mortalities were observed during the surveillance. Bonamia ostreae / B. exitiosa and Marteilia refringens were not detected. Marteilia pararefringens has been observed in Norwegian mussels since 2016 and a targeted surveillance was carried out in 2022. M. pararefringens has so far been detected at eight sites. All Marteilia isolates have been sequenced and typed as Marteilia pararefringens, which is presumed specific for mussels. The work will be continued in 2023, linked to research on the distribution of M. pararefringens in wild mussels. We propose a revision of the surveillance programme and application for disease free status for Bonamia exitiosa in Norwegian flat oysters. We also propose an application for lifting the suspension of free status for Bonamia ostreae in Arendal and Marteilia refringens at Bømlo.
1 - Introduction
The surveillance programme for bonamiosis and marteiliosis in European flat oysters, Ostrea edulis, and Marteilia sp. in blue mussels, Mytilus sp.
The surveillance programme for bonamiosis and marteiliosis in European flat oysters, Ostrea edulis, and blue mussels, Mytilus sp. is carried out by the Institute of Marine Research according to a contract with the Norwegian Food Safety Authority. The over-all aim is to gain knowledge on the health situation of farmed and commercially exploited Norwegian flat oysters and mussels, and maintain the free status for Marteilia refringens and Bonamia ostreae.
There is a connection between farmed and wild populations, due to the geographically widespread and extensive nature of the bivalve industry. Some oyster farmers collect wild seed or half-grown oysters for on-growth in farms. The work therefore requires a tight link between the surveillance programme and the on-going research projects, especially on species distribution and population dynamics, as well as the building of a time-series of data on wild stocks.
After the detection of Marteilia sp. in blue mussels in 2016, IMR increased the effort to study this case, including distribution and parasite life cycle. This report gives a brief overview of the present situation, results from 2022 and the prospects for the work in 2023.
Norwegian flat oyster, Ostrea edulis , are used in re-stocking projects
There is a growing interest in the re-establishment of wild oyster beds in Northern Europe. The restoration of wild oyster beds is dependent on the availability of flat oysters free from Bonamia spp. and Marteilia refringens ( Sas et al . 2020). Both the oyster farming industry and re-stocking projects therefore focus on where to find naïve flat oyster populations that are free from Bonamia spp, as well as other pathogens that may affect the populations. In the present situation – and after the re-occurrence of Bonamia ostreae in Limfjorden, Denmark, in 2014 – safe sources of oysters can only be found in Sweden and Norway. Norwegian populations of European flat oysters have been monitored since 1989 and are considered free from notifiable diseases ( Mortensen et al . 2016; 2020 ). Oysters from Hafrsfjord in Rogaland are being used in re-stocking trials in the North Sea. Oysters from this area are included in the surveillance programme, and the stocks in Hafrsfjord are monitored every 2 years.
Marteilia pararefringens sp. nov. in blue mussels, Mytilus edulis in Norway
The blue mussel, Mytilus spp, populations are changing, and there are numerous reports on an un-explained “disappearance” or mortalities from many Nordic areas. It is not known to which extent diseases play a role in these changes. The wild mussel beds are not monitored on a regular basis, and there is a limited control of mussel farms, using traditional longline cultivation based on the collection of wild seed. Marteilia pararefringens has been found in blue mussels, Mytilus edulis, in five traditional heliothermic marine oyster lagoons, two wild populations and one mussel farm in western Norway (see Figure 1 and report from 2021 ( Mortensen et al . 2022 )). M. pararefringens was regarded as a strain / type / sub-species of M. refringens (Type M), but is now proposed a distinct species, infecting mussels only ( Kerr et al . 2018 ). The effect of M. pararefringens on the mussel populations is the subject of an on-going research project and will be reported and published as the project progresses.
2 - Material and methods
The surveillance and following work were performed according to EU regulations 2016/429, 2017/625 and 2020/689.
Blue mussels are found along the entire Norwegian coast and Spitsbergen. Farming is concentrated in Mid-Norway (Trøndelag and Nordland). Flat oysters are found mainly in Southern Norway, north to Rogaland, with smal, patchy populations north to Romsdal.
Sampling periods were defined according to the periods when the prevalence of Bonamia ostreae and Marteilia sp. (sporulating stage) are highest in the northernmost areas where they have been detected ( Engelsma et al . 2010; Bøgwald et al . 2022). The selected sampling sites are shown in Figure 1 and listed in Table 1. Usually, the surveillance includes an on-site survey, as the state of the population (density, reproduction, signs of mortality) are considered important meta-data.
In 2022, both oysters and mussels were sampled from Arnevik – Kaldvellfjorden in Agder. One company farm oysters and mussels in this area, and combine traditional farming with harvest of wild spat, half-grown and market size oysters from local, wild stocks. Oysters were collected from shallow water at Sauesøya and mussels were collected from a wild population close to a mussel farm in Arnevik. Oysters and mussels were transported to the Institute of Marine Research (IMR) in Bergen. At Rissastrømmen at Fosen, Trløndelag mussels were collected by personnel from the Food Safety Authority and sent to IMR Bergen by over-night mail (Table 1). At Langesand, Agder, the site was inspected by skin diving. Samples were not collected, as this site is no longer included in the surveillance programme.
All oysters and mussels were processed at the IMR laboratory in Bergen, according to standard methodology, and under ISO 17025 QA. The samples, consisting of 152 specimens, were split in two, to be analyzed using histology as well as Polymerase Chain Reaction analysis ( PCR) ( Table 1 ). Histology was performed using dorso-ventral cross sections, fixed in Davidson’s fixative, embedded in paraffin, sectioned at 3 µ m, stained with Hematoxylin Erythrosin Saffron (HES), mounted with a cover slip and observed at 100 to 1000 x magnification. Samples for PCR were fixed in ethanol. DNA was extracted from ethanol fixed digestive gland tissue. Marteilia detection and typing was done by PCR as described by Le Roux et al. (2001).
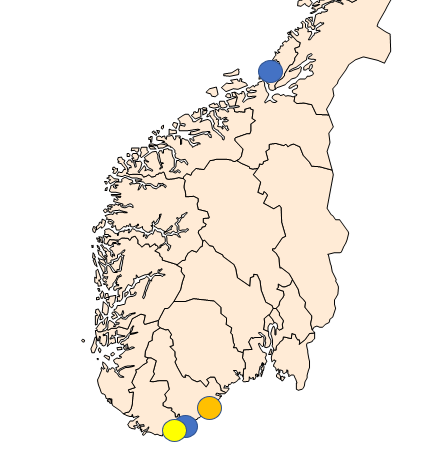
Table 1. Sampling and surveillance of flat oysters ( Ostrea edulis ) and mussels ( Mytilus sp.) in 2022. The flat oyster population at Langesand (orange) was inspected but not sampled in 2022. Sites are shown on the map in Figure 1.
| Sampling site | Oysters | Mussels | |||
| Spring-summer | Autumn | Spring | Autumn | ||
| Sørskjell, Arnevik, Agder | 152 | 152 | 75 by microscopy, 77 by PCR 75 by microscopy, 77 by PCR | ||
| Rissastrømmen, Trøndelag | 152 | 75 obs by microscopy, 77 by PCR | |||
| Langesand, Agder | x | Inspection only | |||
3 - Results
Bonamia spp. or Marteilia refringens were not observed in any sample in 2022. Results from the sites listed in Table 1 are briefly described below.
Examination of flat oysters Langesand ( 58.5392, 8.9376 ) Langesand has a large population of flat oysters. The site was previously included in the surveillance programme and subjected to a targeted Bonamia survey (see previous reports). The site has an apparent stable sub-population of flat oysters growing from 3-6 m depth mostly protected from harvesting and climatic events. From 3 m to surface the population has variable recruitment success and survival due to predation, harvesting and ice conditions. Both sub-populations contain specimens of several year-classes with older specimen dominating in the deeper stratum. There was no sign of elevated mortality. Oysters were not sampled for further examination in 2022.
Arnevik - Kalveldfjorden (58.5253, 8.9108) In the Arnevik area we inspected both mussel and oyster banks from a boat. There was no sign of mortalities in the field. By macroscopic examination the sampled oysters appeared healthy and in good condition. Microscopy revealed gonads in late stage of early gametogenesis, where the follicles were filled with primary oocytes / spermatids. No Bonamia spp. or Marteilia refringens were detected in any of the oyster samples examined under the microscope. Ciliates were observed in eight individuals; one individual was infected with rickettsia - like organisms (RLO). Three individuals had inflammation (hemocyte infiltration) in the digestive gland and gonads.
Examination of Mussels
Strømmen, Rissa, Trøndelag ( 63.5777, 9.9404 ) Mussels from Rissastrømmen come from a wide tidal current used as intermediate grow-out and harvest of farmed mussels. The mussels ranged from 44 to 89 mm, with a mean length of 65 mm. Macroscopically the mussels seemed healthy, with few deformities or other abnormalities. 75 mussels were examined by microscopy. All but one mussel was in good condition. An intracellular bacterial colony was observed in one individual. No other abnormal findings were observed.
Arnevik - Kalveldfjorden ( 58.5253, 8.9108) The mussels were collected on shallow water near a site with farming of both mussels and oysters. Green pigmentation (presumably Coccomyxa -infections) was observed in the mantle and mussel tissue of several mussels. 75 mussels were examined by microscopy. The mussels were in varying condition. Local infiltration in the mantle was observed in several individuals and large granulomas were observed in some individuals. Several intracellular bacterial colonies were observed in one individual. One hermaphrodite was observed.
4 - Discussion and conclusions
The health status of Norwegian flat oysters
Marteilia refringens has never been detected in Norway, and Norwegian flat oysters, O. edulis, appear free from Bonamia spp.
Healthy flat oysters are a valuable resource. It is important to monitor Norwegian stocks and disseminate the information on their health status to obtain a consensus on how to protect and care for this resource. We are monitoring Norwegian flat oyster populations and aim at using the data gained in a Nordic and European context. The monitoring of stocks is linked to the national health surveillance, and through the contact with European scientists to both genetic studies ( Margen project) and re-stocking programmes ( https://noraeurope.eu/ ).
There is a small commercial harvest of wild flat oysters along the south and south-western coast of Norway. During this harvest, half-grown oysters are sometimes collected and used as seed in oyster farms. One example is Sørskjell Ltd, located in Arnevik, which were included in the surveillance in 2022. Here, the shellfish farmer produces both mussels and oyster in suspended culture and uses local beds of oysters both as a bottom culture and a source of half-grown oysters for the farm. The production is efficient but demonstrate the link between wild and farmed oysters.
Examination of mussels
Marteilia pararefringens has been found in blue mussels, Mytilus sp., in five traditional heliothermic marine oyster lagoons, two wild populations and one mussel farm in western Norway (see Figure 1 and report from 2021). These findings indicate that M. pararefringens is not restricted to the heliothermic lagoons. We have started a survey including several of the traditional oyster lagoons as well as mussels in closed, sheltered areas. To optimize the sampling regime in the surveillance programme and the Marteilia study, we collaborate with all active mussel producers to map the farms, spat collection areas and translocations.
Most of the Norwegian mussel production is in Trøndelag and Nordland. All farmed mussels from this (large) area are processed, packed, and distributed by two dispatch centers, owned by Norgeskjell. Although two selected sites (close to the dispatch centres) are included in the surveillance programme. The dispatch centers will be followed up and their sources of mussels mapped. It is important to establish a surveillance model for this area and initiate a targeted sampling.
Based on our findings, M. pararefringens does not seem to infect flat oysters. It is however important to ensure that oysters do not act as vectors. Between 2016 and 2022 we have sampled 29 sites to map the distribution of M. pararefringens in Norwegian mussel populations. M. pararefringens is so far detected at eight sites. In 2023, we will collect oysters from these sites (if present) and perform a targeted search for M. pararefringens in oyster tissues.
Conclusions and recommendations
The surveillance will be continued according to the plan discussed with the Norwegian Food Safety Authority, and with the aim of obtaining a time series of data on the health status of flat oysters and mussels along the Norwegian coast.
The surveillance programme will be focused on mussel and oyster farms and/or populations that are commercially harvested for on-growth, transport to the markets or used in re-stocking programmes.
The detection of Marteilia pararefringens in mussels in new areas indicate that this parasite is more widely distributed than previously known. The geographical survey will be continued during summer 2023.
The health surveillance is linked to monitoring of stocks, oyster genetic studies and European oyster projects, strengthening the scientific basis for a strong and adequate management of the few remaining, healthy flat oyster populations in Europe.
Flat oysters do not appear susceptible to M. pararefringens. Scientific studies focus on understanding the infection and life cycle of M. pararefringens in mussels, as a background for a risk assessment and an epidemiological study of the spreading of this parasite in North European mussel populations. Flat oysters will be sampled in areas with M. pararefringens to obtain more data on the status of the oysters.
We consider Norwegian populations of flat oysters as free from Bonamia spp. and Marteilia refringens. We advise the Norwegian Food Safety Authority to apply for disease free status for B. exitiosa in Norwegian flat oysters, and a lifting of the suspension of free status for Bonamia ostreae in Arendal. If additional samples are required to fulfil the criteria, this should be included in the sampling plan before May 2023.
Acknowledgements
Thanks to Per Christian H. Lund for assistance during sampling.
5 - References
Bøgwald M, Skår CK, Karlsbakk E, Alfjorden A, Feist SW, Bass D, Mortensen S (2022). The infection cycle of Marteilia pararefringens in blue mussels Mytilus edulis in a heliothermic marine oyster lagoon in Norway. Diseases of Aquatic Organisms 148: 153–166, https://doi.org/10.3354/dao03651
Commission Delegated Regulation (EU) 2020/689 of 17 December 2019 supplementing Regulation (EU) 2016/429 of the European Parliament and of the Council as regards rules for surveillance, eradication programmes, and disease-free status for certain listed and emerging diseases. http://data.europa.eu/eli/reg_del/2020/689/oj
Engelsma MY, Kerhoff S, Roozenburg I, Haenen OLM, van Gool A, Sistermans W, Wijnhoven S, Hummel H (2010). Epidemiology of Bonamia ostreae infecting European flat oysters Ostrea edulis from Lake Grevelingen, The Netherlands. Marine Ecology Progress Series 409: 131 – 142.
Kerr R, Ward GM, Stentiford GD, Alfjorden A, Mortensen S, Bignell JP, Feist SW, Villalba A, Carballal MJ, Cao A, Arzul I, Ryder D, Bass D (2018). Marteilia refringens and Marteilia pararefringens sp. nov. are distinct parasites of bivalves and have different European distributions. Parasitology 1–10. https://doi.org/10.1017/ S003118201800063X
Le Roux F, Lorenzo G, Peyret P, Audemard C, Figueras A, Vivares C, Gouy M, Berthe F (2001). Molecular evidence for the existence of two species of Marteilia in Europe. Journal of Eukaryote Microbiology 48: 449-454.
Mortensen S, Skår C (2020). The surveillance and control programme for bonamiosis and marteiliosis in European flat oysters, Ostrea edulis , and blue mussels, Mytilus sp. in Norway in 2019. Rapport fra havforskningen nr 36, 2020, 16 s. https://www.hi.no//hi/nettrapporter/rapport-fra-havforskningen-en-2020-36
Mortensen S, Skår CK, Harkestad LS (2013). Hemisk neoplasi hos norsk flatøsters, Ostrea edulis . Norsk Veterinærtidsskrift 125 (7): 438 -442.
Mortensen S, Sælemyr L, Skår CK, Bodvin T, Jelmert A (2016). Health surveillance of the flat oyster populations in Aust-Agder County, southern Norway in the period 2009 – 2015. Rapport fra havforskningen nr 11, 2016, 11s.
Mortensen S, Skår C, Sælemyr L (2020). Summarizing the screening for Bonamia ostreae in Norwegian populations of flat oysters, Ostrea edulis . Rapport fra Havforskningen no 24, 2020. ISSN:1893-4536, https://www.hi.no/hi/nettrapporter/rapport-fra-havforskningen-en-2020-24
Regulation (EU) 2016/429 of the European Parliament and of the Council of 9 March 2016 on transmissible animal diseases and amending and repealing certain acts in the area of animal health (‘Animal Health Law’). https://eur-lex.europa.eu/eli/reg/2016/429/oj
Regulation (EU) 2017/625 of the European Parliament and of the Council of 15 March 2017 on official controls and other official activities performed to ensure the application of food and feed law, rules on animal health and welfare, plant health and plant protection products. http://data.europa.eu/eli/reg/2017/625/oj
Sas H, Deden B, Kamermans P, zu Ermgassen P S E, Pogoda B, Preston J, Helmer L, Holbrook Z, Arzul I, van der Have T, Villalba A, Colsoul B, Lown A, Merk V, Zwerschke N, Reuchlin E (2020). Bonamia infection in native oysters ( Ostrea edulis ) in relation to European restoration projects. Aquatic Conservation. Marine and Freshwater Ecosystems 30: 2150-2162. https://doi.org/10.1002/aqc.3430