Havforskningsinstituttet har undersøkt prevalensen av infeksiøs lakseanemivirus (ISAV), Salmonid alfavirus (SAV, PD-virus), piscine ortoreovirus 1 (PRV-1), piscine myokardittvirus (PMCV) og infeksiøs pankreasnekrosevirus (IPNV) ) infeksjoner i vill atlantisk laks postsmolt fanget i 2021 i tre fjordsystemer lokalisert i tre akvakulturproduksjonsområder (PO2, 3 og 5). Fisken ble samlet inn som en del av det nasjonale overvåkingsprogrammet for lakselus i ytre deler av Boknafjord (N = 50), Hardangerfjord (N = 57) og Romsdalsfjord (N = 73) ved tråling i perioden mai-juni. SAV ble påvist i 2 postsmolt fra Romsdalsfjord. PRV-1 ble påvist i 11 (6 %) av postsmolten (8 fra Boknafjord, 1 fra Hardangerfjord og 2 fra Romsdalsfjord). Lave konsentrasjoner av PMCV-RNA ble påvist i 19 (11 %) postsmolt (13 fra Boknafjord og 6 fra Romsdalsfjord). ISAV og IPNV ble ikke påvist i noen av fisken. Generelt hadde virus-positive fiskene høye Ct-verdier som kan tyde på svært lave viruskonsentrasjoner. Resultatene i den nåværende rapporten viste at postsmolt fra fjorder hadde svært lav forekomst av virussmitte som er vanlig i norsk havbruk. Disse funnene utfyller og bekrefter de tidligere rapporterte dataene våre og kan tyde på at prevalensen av virusinfeksjoner i villaks postsmolt ikke påvirkes signifikant av forekomsten av disse infeksjonene i fiskeoppdrett.
Annual report on health monitoring of wild anadromous salmonids in Norway 2022
— Screening of Atlantic salmon (Salmo salar) postsmolts from Boknafjorden, Hardangerfjorden and Romsdalsfjorden for viral infections
Rapportserie:
Rapport fra havforskningen 2023-41
ISSN: 1893-4536
Overvåkingsgruppens rapporter
Publisert: 03.10.2023
Prosjektnr: 15697-01
Oppdragsgiver(e): Norwegian Food Safety Authority
Forskningsgruppe(r):
Smittespredning og sykdom
Program:
Miljøeffekter av akvakultur
Godkjent av:
Forskningsdirektør(er):
Geir Lasse Taranger
Programleder(e):
Terje Svåsand
English summary
Sammendrag
The Institute of Marine Research has investigated the prevalence of infectious salmon anaemia virus (ISAV), salmonid alphavirus (SAV, PD-virus), piscine orthoreovirus 1 (PRV-1), piscine myocarditis virus (PMCV) and infectious pancreatic necrosis virus (IPNV) infections in migrating wild Atlantic salmon postsmolts captured in 2021 in three fjord systems located in three aquaculture production areas (PO2, 3 and 5). The fish were collected as part of the national salmon lice monitoring program in the outer parts of Boknafjord (N = 50), Hardangerfjord (N = 57) and Romsdalsfjord (N = 73) by trawling during the period May-June. SAV was detected in 2 postsmolts from Romsdalsfjord. PRV-1 was detected in 11 (6%) of postsmolts (8 from Boknafjord, 1 from Hardangerfjord and 2 from Romsdalsfjord). Low concentrations of PMCV-RNA were detected in 19 (11 %) of postsmolts (13 from Boknafjord and 6 from Romsdalsfjord). ISAV and IPNV were not detected in any of the test fish. Generally, the virus-positive fish had high Ct-values which may indicate very low virus concentrations. The results in the current report showed that migrating postsmolts from fjords had a very low occurrence of virus infections common in Norwegian aquaculture. These findings complement and corroborate our previously reported data and may suggest that prevalence virus infections in wild salmon postsmolts is not significantly influenced by the occurrence of these infections in fish farming.
1 - Introduction
Viral diseases in Atlantic salmon farming in Norway is a serious problem which has an impact on welfare of infected salmon and often leads to substantial economic losses (Table 1) [1]. The most common viral diseases in salmon farming are pancreas disease (PD), caused by salmonid alphavirus (SAV), infectious salmon anaemia (ISA), caused by ISA virus (ISAV), heart and skeletal muscle inflammation (HSMI), caused by piscine orthoreovirus 1 (PRV-1), cardiomyopathy syndrome (CMS), caused by piscine myocarditis virus (PMCV) and infectious pancreatic necrosis (IPN), caused by IPN virus (IPNV).
PD is one of the major diseases in fish farming and much of the fish in endemic areas are believed to become infected with SAV through a production cycle. In Norway, SAV3 was the only subtype detected in salmon farming for a long time, but in 2010 SAV2 was also detected in farmed salmon and since then, PD outbreaks have been dominated by SAV2 in central Norway and SAV3 in western Norway [1,2].
ISA is a serious disease that has led to severe epizootics in aquaculture in Faroe Islands, Norway and Chile, with enormous economic consequences for industry. Since the 1993, there have been relatively few ISA outbreaks in Norway. However, an increase in the number of ISA outbreaks in recent years along the entire Norwegian coast indicates a growing problem in the salmon farming. There are two variants of the virus, one that causes disease (virulent, HPR-del) and one that does not cause disease (avirulent, HPR-0). There are growing evidence that virulent ISAV arises from avirulent ISAV, but what triggers this is not yet known [3].
Heart and skeletal muscle inflammation (HSMI) is the most abundant viral disease in salmon farming. The disease is an increasing problem in fish farming in Norway with 79–188 annual registered cases of HSMI in recent years [1,4]. High PRV-1 viral loads are found in fish developing HSMI but may also occur in healthy fish.
Cardiomyopathy syndrome (CMS) is a growing problem in Norwegian salmon farming with 82 to 155 annual outbreaks in the last 5 years [1,5].
Infectious pancreatic necrosis (IPN) was one of the major serious viral diseases in Norwegian salmon farming in the 1980s. However, the improvement of routines at hatcheries and the use of IPNV-resistant (QTL) salmon strains led to significant reduction in the numbers of IPN-outbreaks [1].
|
2017 |
2018 |
2019 |
2020 |
2021 |
|
|---|---|---|---|---|---|
|
PD |
176 |
163 |
152 |
158 |
100 |
|
ISA |
14 |
13 |
10 |
23 |
25 |
|
HSMI |
93 |
104 |
79 |
161 |
188 |
|
CMS |
100 |
101 |
82 |
154 |
155 |
|
IPN |
23 |
19 |
23 |
22 |
20 |
|
Total |
368 |
406 |
400 |
346 |
518 |
Pathogen exchange between farmed and wild salmon occurs, e.g. salmon lice. Hence, disease outbreaks in salmon farms may lead to increased infection pressure on wild fish populations. There is an increasing public concern of this negatively impacting wild salmonids in Norway. However, although the amount of the data and knowlegde on the prevalence of virus infections in wild salmonid populations in Norway is increasing [6,7], but is still limited. Furthermore, it is difficult to quantify disease incidence and its impact on wild fish since sick individuals may be less catchable or may disappear in nature unnoticed (e.g. due to predation). Therefore, it is challenging to evaluate the impact of pathogens on individuals as well as stocks, since we normally are only able to collect infected but non-diseased fish such as individuals that has recently acquired or has survived an infection (carriers). To increase our knowledge, long term surveillance programmes creating a timeseries with sufficient data are necessary.
The effect of fish farming on the infection status of wild salmon stocks may be evaluated by comparing pathogen prevalence in wild fish populations originating from areas having different fish farming intensities and disease outbreak profiles.
Wild salmon may be infected by viruses prevalent in salmon farming; in rivers as fry or parr by virus-infected farmed escapees and spawning wild salmon, or from salmon farms in the fjord when migrating as smolts or returning as adults. Therefore, infection status in migrating smolts may represent a direct indicator of infection pressure from salmon farming during their migration routes. However, the study of viral infection during all life stages of salmon life cycle is necessary to assess the overall impact of diseases in fish farming on the salmon wild stocks.
Since 2012, the Institute of Marine Research (IMR) has been commissioned by the Norwegian Food Safety Authority (NFSA) to carry out an annual health monitoring of wild anadromous salmonids in Norway. The current monitoring activities are financed by both NFSA and the Norwegian Ministry of Trade, Industry and Fisheries (NFD). The activities lie within a prioritized research area at IMR which addresses the environmental impact of disease transmission from Norwegian fish farming to wild fish. The surveillance activities aim to evaluate the virus transmission from farmed fish to wild salmonids by monitoring and identifying changes in the prevalence of selected viruses in wild salmonids as a result of fish farming activities. In addition, the surveillance aims to increase the knowledgebase about pathogens in wild salmonids in general, as well as establish a biobank that can be used when new disease challenges arise. Furthermore, the surveillance consolidates with the other activities in the larger strategic research effort on diseases and disease transmission in wild fish.
Part of the research activities in the surveillance program aims to generate data about:
- Virus prevalence in fry, parr, postsmolt and returning adult salmon
- Prevalence of viruses in sea trout
- Prevalence of infections in escaped farmed salmonids
- Genotypes and characteristics of detected viruses
The virus screening is based on selected materials obtained through monitoring of virus infections in wild salmonids project and other associated projects at IMR, such as:
- National salmon lice monitoring program (NALO)
- National escaped salmon monitoring program
- Etne research station (fish trap)
- Dale research project
- Atlantic salmon at sea (Seasalar) project
- CoASal project
The current monitoring program aims to investigate the occurrence of virus infections in wild salmonids captured from different Norwegian coastal areas with different farming intensities and disease outbreak frequencies. Each year selected sets of fish are analysed in order to complement or complete our data and time series. Part of the results from pathogen screening are used in an annual health monitoring of wild anadromous salmonids in Norway commissioned by NFSA. The generated knowledge from the program contributes to the institute's main goal/strategy in providing advice and further development of sustainable management of aquaculture and is utilized in the IMR's annual risk assessment of Norwegian fish farming.
For 2022, the OK program investigated the occurrence of ISAV, SAV, PRV-1, PMCV and IPNV infections in migrating postsmolt from the Boknafjorden, Hardangerfjorden and Romsdalsfjorden. The three selected fjords are located in three different salmon production areas (PO) that have different farming intensities and disease profiles and were therefore selected based on an assessment of the risk of infection by these viruses [6,7]. Fish from each site were analyzed for virus infections using real-time RT-PCR.
2 - Aim
The aim of the current study was to investigate the occurrence of SAV, ISAV, PRV-1, PMCV and IPNV infections in migrating wild Atlantic salmon postsmolts captured in 2021 in three fjord systems located in three aquaculture production areas (PO2, PO3 and PO5) with differing risk for virus infection of wild fish.
3 - Materials and methods
To provide data about the prevalence of different viruses in different salmon life stages and different geographical regions, we selected fish from our available material from 2021 for the analysis. These materials were migrating postsmolts captured, as part of the national salmon lice monitoring program (NALO) [8], in the outer parts of the Boknafjorden, Hardangerfjorden and Romsdalsfjorden by trawling during the period May-June 2021 (Fig. 1 and Table 2).
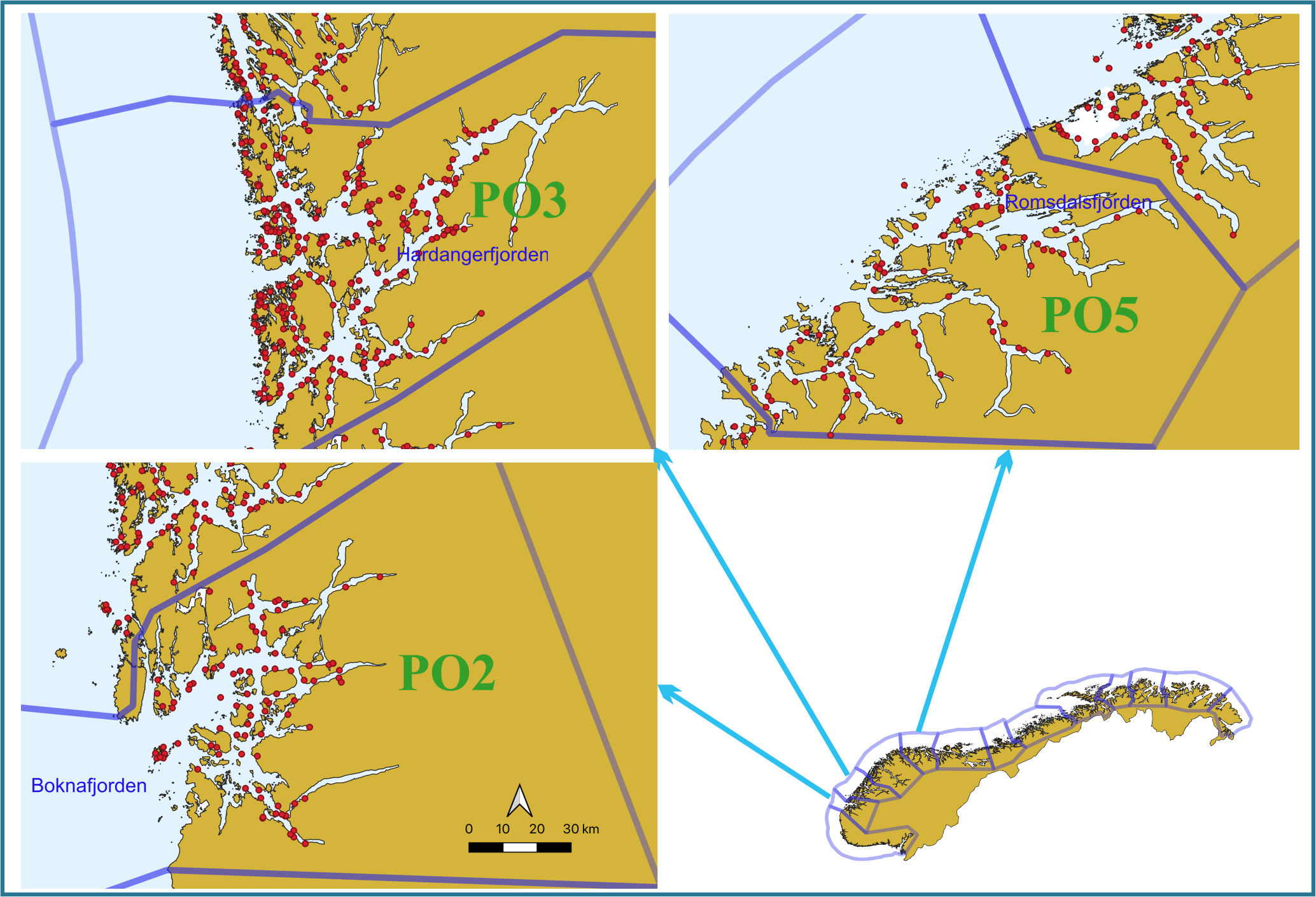
Weight and length of all postsmolts were recorded and the fish were then frozen (-20 oC) as soon as possible. At autopsy, tissues from the gills, head kidney and heart were taken from the fish while still frozen and stored at -80 oC. Samples for analysis were sent on dry ice to an accredited commercial laboratory for RNA extraction and virus testing (Pharmaq Analytiq AS; https://www.pharmaq.com/en/analytiq). All fish were tested for SAV, ISAV, PRV-1, PMCV and IPNV (Table 2) by real-time RT-PCR assays (for detection of viral RNA). A total of 900 analyses were performed on 180 fish and included in the current report.
4 - Results
Low concentrations of SAV-RNA were detected in two postsmolts from Romsdalsfjorden, whilst ISAV was not detected in any of the tested fish (Table 2). PRV-1 was detected in 11 (6%) of postsmolts. The PRV1-positive fish were from Boknafjorden (N=8) and Romsdalsfjorden (N=3). The positive fish, except two, had high ct-values (35-39) which is likely to represent weak or false positive results. On the other hand, two fish had ct-values of 27 suggesting a moderate virus concentration. Low concentrations (ct-values 34-39) of PMCV were detected in 19 (11%) fish. The low ct-values suggest a weak or false positive results. The PMCV-positive post smolts were collected from Boknafjorden and Romsdalsfjorden. IPNV was not detected in any of the tested fish.
|
Collection Site (production area) |
Number |
SAV |
ISAV |
PRV-1 |
PMCV |
IPNV |
|---|---|---|---|---|---|---|
|
Boknafjorden (PO2) |
50 |
0 |
0 |
8 (16%) |
13 (26%) |
0 |
|
Hardangerfjorden (PO3) |
57 |
0 |
0 |
1 (2%) |
0 |
0 |
|
Romsdalsfjorden (PO5) |
73 |
2 (3%) |
0 |
2 (3%) |
6 (8%) |
0 |
|
Total |
180 |
2 (1%) |
0 |
11 (6%) |
19 (11%) |
0 |
5 - Discussion and conclusion
Low concentrations of SAV were detected in 2 migrating postsmolt from production area PO5. PD is prevalent (endemic) in the three collection areas (PO2, PO3 and PO5). Therefore, it is likely that migrating postsmolt were exposed to SAV released from the fish farms. However, the very low concentrations (Ct-values: 35 and 36) was just below the detection limit of the PCR assay (cut-off 37) and makes to difficult to conclude if the results indicating weak or false positive. The low prevalence of SAV infections in the tested migrating smolt is consistent with previous findings in wild salmonids [6,7]. Our earlier report showed that migrating smolts from Trondheimsfjorden which has no fish farming activities also had very low occurrence of SAV infection [9]. Similarly, very low prevalence of SAV was detected in returning adult salmon, postsmolt at Norwegian sea or juveniles from Norwegian rivers [6,7].
ISAV and IPNV were not detected in any of the tested fish. There was only one ISA and very few IPN outbreaks in the three fjords during the postsmolt migration period and the exposure to these viruses has therefore been considered to be very low [1,7].
PRV-1 infection is abundant in fish farming in production areas PO2-13. We detected the virus in 11 postsmolts from PO2, PO3 and PO5 (Table 2). Two the postsmolts had moderate virus concentrations whilst 9 fish had concentrations at the detection limit of the assay. PRV-1 has been detected in wild salmon and sea trout by real-time rt-PCR [10-12]. Reports has shown that there was no regional pattern in virus genotypes isolated from wild and farmed salmon, suggesting prolonged and extensive spread due to aquaculture activities (fish transport) and frequent exchange of the virus types between farmed and wild fish [12,13]. However, little is known about the mechanism of transmission of the virus.
PMCV was detected at low concentrations (ct-values 35-39) in 19 (10%) of the tested fish.
The ct-values of the RT-PCR is likely to indicate false positive or very low virus concentrations. Previous reports have shown low prevalence of PMCV infections in wild salmon and that infection was not associated with fish farming activities [6,14,15].
The farming intensities in PO2, PO3, and PO5 were 42.4, 53.7 and 23.7 tonne/km2 respectively [7]. Hardangerfjorden is located in PO3 which is one of the areas with the highest fish farming intensities in Norway. There was only one virus-infected (PRV1-positive) postsmolt collected from this fjord suggesting that fish farming intensity was not associated with an increased occurrence of infection with the tested viruses in wild fish.
The current findings are in line with our previous reports that showed no apparent relationship between the prevalence of virus infection in wild salmon and the fish farming intensity or the frequency of disease outbreaks in collection areas [7,9,11,12]. These observations may indicate that wild salmon are exposed to a low infection pressure from fish farming. However, the possibility that infection may lead to rapid disappearance or altered behaviour of the infected fish, and therefore may affect the results, cannot be ruled out. Other explanation for the low prevalence of viruses in postsmolts is the time needed after virus exposure (incubation time) before the virus can be detected in tissues of fish. To verify our observations, a large PD-vaccine smolt release study was therefore conducted in 2018 and 2019. In this study, a total of 52 000 (28 000 PD-vaccinated and 24 000 control) smolt were released in rivers Etneelva and Daleelva located in production areas (PO3 and PO4) which had a high number of PD cases during the release period. The survival rate of returning adult salmon in the subsequent years were determined in both vaccine and control groups and used to estimate the mortality that may be attributed to SAV infection from fish farming in release areas. The results (unpublished) from the study did not show any statistically significant differences in the mortality rate between the vaccine and the control groups and therefore support our observations that infection pressure of SAV from fish farms to wild salmon is low. Furthermore, screening smolt used in sentential cages placed in PO3 for a period of 2 weeks for ISAV and SAV infections did not reveal virus infection in these smolt.
The results in the current report showed migrating postsmolts from fjords had a very low occurrence of infection by 5 viruses prevalent in Norwegian aquaculture. These findings complement and corroborate our previously reported data and may suggest that prevalence of virus infections in wild salmon at different life stages are not significantly influenced by the occurrence of these infections in fish farming. It is also suggesting that it is unlikely that wild salmon is the major reservoir that spill over these viruses to fish farming. There are still significant gaps in our knowledge about diseases in wild fish and the interaction between farmed and wild fish [6,7]. Time series of samples of all life stages of wild salmonids from areas with different salmon farming intensities are necessary to better evaluate and understand the long-term effect of infection pressure from aquaculture on the virus prevalence in wild salmon populations. Such series will also enable us to assess changes in the prevalence due to increased fish farming activities, increased pathogen virulence, the emergence of new diseases and climate change.
6 - References
- Sommerset, I., et al. (2022). Fish health report 2021 (in Norwegian). Norwegian Veterinary Institute, 2a/2022. Available at https://www.vetinst.no/rapporter-og-publikasjoner/rapporter/2022/fiskehelserapporten-2021
- Hjortaas, M. J., et al. (2013). “The first detections of subtype 2-related salmonid alphavirus (SAV2) in Atlantic salmon, Salmo salar L., in Norway”. Journal of Fish Diseases, 36: 71-74.
- Rimstad, E., and Markussen, T. (2020). Infectious salmon anaemia virus-molecular biology and pathogenesis of the infection. Journal of Applied Microbiology, 129:85-97.
- Løvoll, M., et al. (2012). “Quantification of piscine reovirus (PRV) at different stages of Atlantic salmon Salmo salar production”. Diseases of Aquatic Organisms, 99: 7-U5.
- Jensen, B. B., et al. (2013). “Risk factors for cardiomyopathy syndrome (CMS) in Norwegian salmon farming”. Diseases of Aquatic Organisms, 107: 141-150.
- Taranger, G. L., et al. (2015). “Risk assessment of the environmental impact of Norwegian Atlantic salmon farming”. ICES Journal of Marine Science, 72: 997-1021.
- Grefsrud, E. S., et al. (2023). “Risk assessment of Norwegian fish farming”. Rapport fra havforskningen 2023-6: Institute of Marine Research, Bergen. Available at https://www.hi.no/hi/nettrapporter/rapport-fra-havforskningen-2023-6
- Nilsen, R., et al. (2017). Lakselusinfestasjon på vill laksefisk langs norskekysten i 2016; med vekt på modellbasert varsling og tilstandsbekreftelse. Rapport fra Havforskningen 1-2017: Institute of Marine Research, Bergen
- Madhun, A. S., et al. (2019). Annual report on health monitoring of wild anadromous salmonids in Norway 2018; Screening of migrating Atlantic salmon (Salmo salar) postsmolts from the Trondheim fjord for viral infections. Rapport fra Havforskningen 2019-28: Institute of Marine Research, Bergen. 9 pp. Available at https://www.hi.no/en/hi/nettrapporter/rapport-fra-havforskningen-en-2019-28
- Garseth, Å. H., et al. (2013). “Piscine reovirus (PRV) in wild Atlantic salmon, Salmo salar L., and sea-trout, Salmo trutta L., in Norway”. Journal of Fish Diseases, 36: 483-493.
- Madhun, A. S., et al. (2016). Occurrence of salmonid alphavirus (SAV) and piscine orthoreovirus (PRV) infections in wild sea trout Salmo trutta in Norway. Diseases of Aquatic Organisms, 120:109-113.
- Madhun, A. S., et al. (2018). “Prevalence of piscine orthoreovirus and salmonid alphavirus in sea-caught returning adult Atlantic salmon (Salmo salar L.) in northern Norway”. Journal of Fish Diseases, 41: 797-803.
- Garseth, Å. H., et al. (2013). “Phylogenetic evidence of long distance dispersal and transmission of piscine reovirus (PRV) between farmed and wild Atlantic salmon”. PloS One, 8: e82202.
- Garseth, Å. H., et al. (2012). “Piscine myocarditis virus (PMCV) in wild Atlantic salmon Salmo salar”. Diseases of Aquatic Organisms, 102: 157-161.
- Madhun, A. S., et al. (2018). “Annual report on health monitoring of wild anadromous salmonids in Norway 2017; Health monitoring of returning adult salmon from river Etne, western Norway ”. Rapport fra Havforskningen 26-2018: Institute of Marine Research, Bergen. 13 pp. Available at https://www.hi.no/en/hi/nettrapporter/rapport-fra-havforskningen-en-2020-16